 
Every circular orbit will have a certain amount of fixed energy and these circular orbits were termed orbital shells. The orbits are termed as stationary orbit. If the atom receives energy from an outside source, it is possible for the electron to move to an orbit with a higher n value and the atom is now in an excited electronic state (or simply an excited state) with a higher energy. Electrons revolve around the nucleus in a fixed circular path termed orbits or shells or energy level. The electronic configuration of Oxygen is He 2s 2 2p 4. The information that we can derive from the above-mentioned Oxygen box is as follows: The atomic number of Oxygen is 8. When the electron is in this lowest energy orbit, the atom is said to be in its ground electronic state (or simply ground state). Learn about the Bohr model of the hydrogen atom and the physics behind it. The oxygen atom belongs to the 16 th group of the periodic table. On the qualitative level these attempts met with some success, and a general picture of electrons occupying orbits in successive levels and sub-levels, similar to that shown in Figure 5.2, began to emerge. 
They also help us explain and predict the behavior of atoms. An atom is a building block of matter that cannot be broken apart using any chemical means. Bohr’s success with the hydrogen atom soon led to attempts both by him and by others to extend the same model to other atoms. Models help us visualize atomic structure. 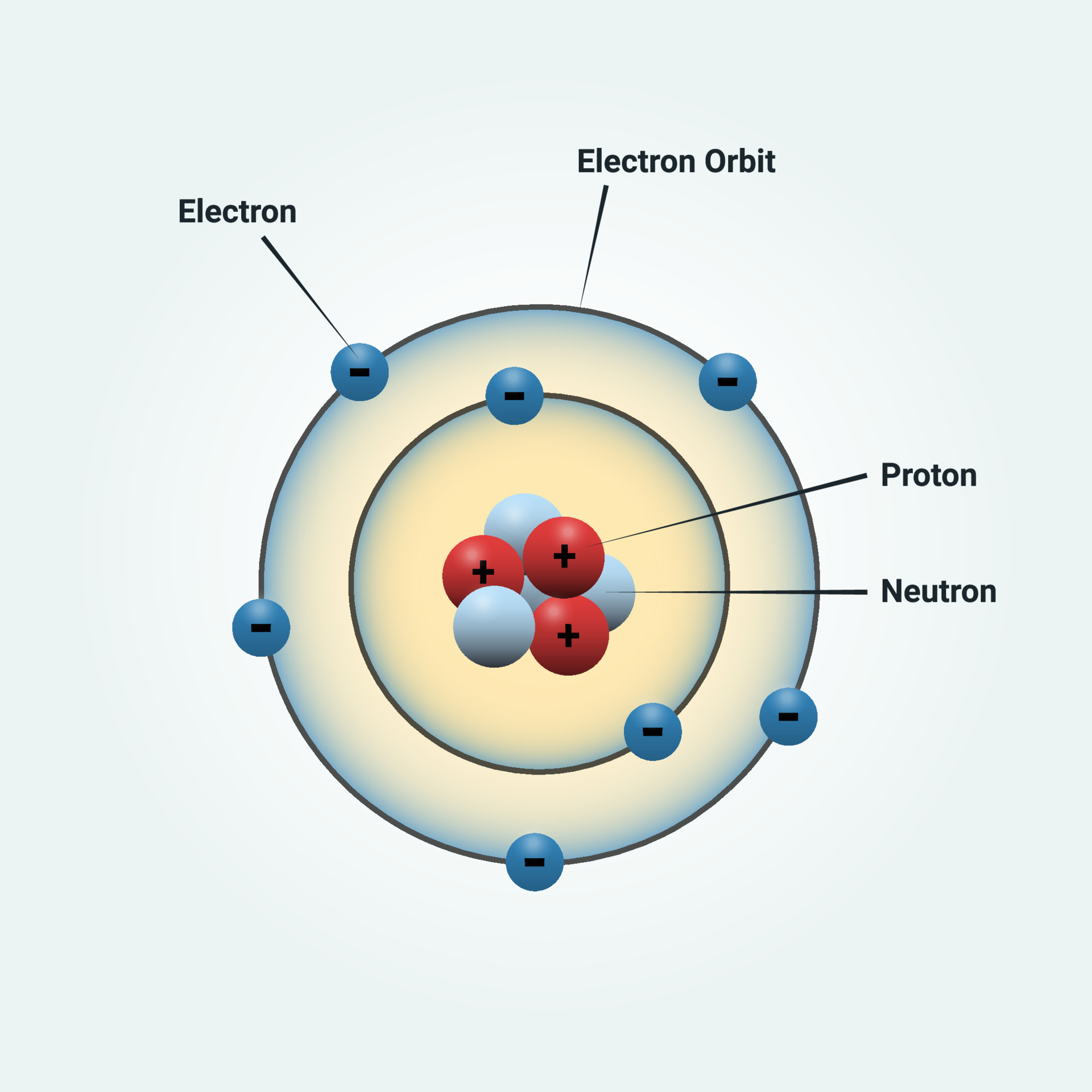
Atoms are way too small to see with the naked eye (and even most microscopes). The law of conservation of energy says that we can neither create nor destroy energy. We can relate the energy of electrons in atoms to what we learned previously about energy. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. The more negative the calculated value, the lower the energy. In 1913, a Danish physicist, Niels Bohr (18851962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Thus, the electron in a hydrogen atom usually moves in the n = 1 orbit, the orbit in which it has the lowest energy. The Bohr model Google Classroom Learn how Bohr models are used to represent atoms. 1: Quantum numbers and energy levels in a hydrogen atom. One of the fundamental laws of physics is that matter is most stable with the lowest possible energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
